Newsletter June 2022

Research updates |
|
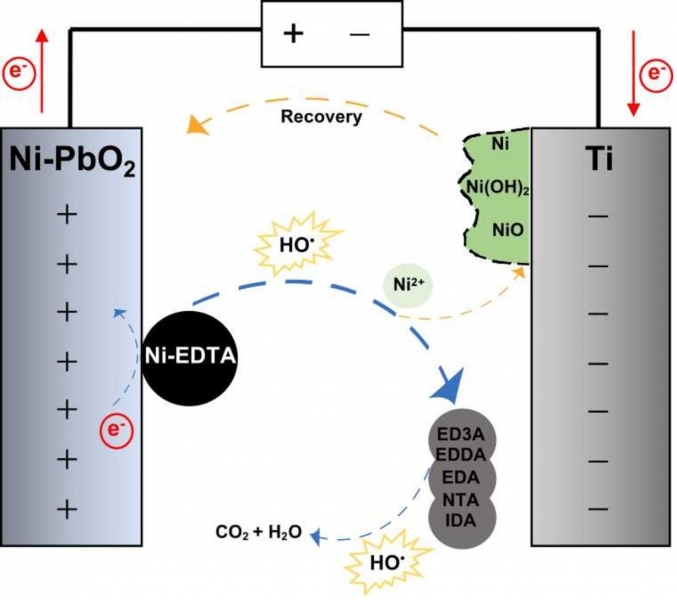
Project 1. The Electrochemical Advanced Oxidation Processes (EAOPs) team have made some progress in the treatment of metal plating wastewater with particular attention given to degradation of Ni-EDTA complexes and recovery of Ni. A pilot-scale EAOP reactor has been fabricated using innovative electrodes designed by CTET. The CTET-designed pilot-scale system can successfully remove 80% EDTA and recover 80% Ni from the Ni-EDTA wastewater at a treatment capacity of 0.9 tonnes/day. |
 |
 |
|
Project 2. The Membrane Capacitive De-ionization (MCDI) team have fabricated a 100-pair electrode module which successfully reduced the conductivity in synthetic groundwater by 60-70% with a water recovery of 86% in a 50-cycle test. The treated groundwater can meet the conductivity recommended in the Australian drinking water guidelines. |
 |
 |
China Learning Centre
While borders are now open, the Yixing CLC is continuing to operate with activities such as an online workshop held on 1st June to assist students to utilize the resources of the UNSW library. Additionally, the CLC is continuing to assist students in finding industrial training placements and to provide a place for undergraduate and postgraduate students to mix with their peers in both a convenient study environment and friendly social setting. |
External communication |
|
Prof. David Waite, the Executive Chairman of CTET and Dr Yuan Wang, the General Manager of CTET met multiple industrial and governmental partners in Beijing, Nanjing as well as Yixing to explore future cooperation opportunities during their recent 10-week visit to China.
|
Publications |
|
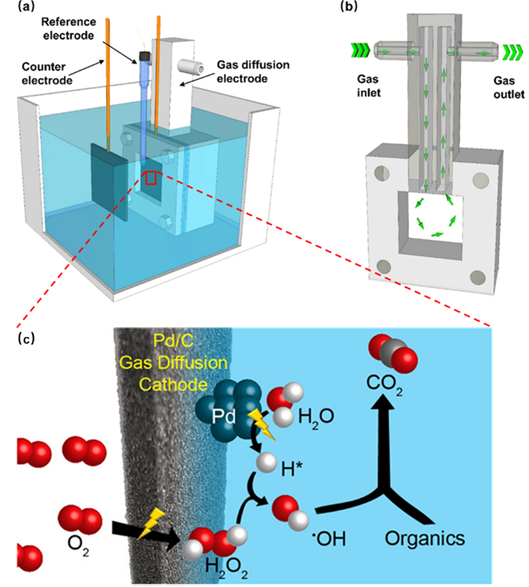
2. Another research paper entitled “Hydroxyl Radical Production via Reaction of Electrochemically Generated Hydrogen Peroxide and Atomic Hydrogen: An Effective Process for Contaminant Oxidation?” has been published in Environmental Science and Technology by CTET. This paper describes investigations of a so-called “green” Fenton technology which requires input of water, air and electricity only. A palladium-coated carbon-PTFE gas diffusion electrode (Pd/C GDE) was used as a catalytic cathode with hydroxyl radical (•OH) formed as a result of the reaction of electrogenerated hydrogen atoms (H*) with in situ generated hydrogen peroxide (H2O2). The studies show that H* and H2O2 (and thus •OH) can be electrogenerated effectively over a wide range of pH (3.2–9.0). These results suggest that by in situ generation of H* and H2O2, the H*/GDE process is able to produce significant amounts of •OH without external chemical addition though further research is needed to increase the durability of the electrodes used. |

 UNSW Sydney launched its first China Learning Centre (CLC) in T3 2020 at the Yixing Industrial Park for Environmental Science and Technology (ES&TP), where UNSW Centre for Transformational Environmental Technologies (CTET) is located, to support those students who cannot return to the campus in Sydney to continue their studies. From T3 2020 to T1 2022, the Yixing CLC has assisted more than 420 students to take online courses, develop thesis projects and carry out industrial training.
UNSW Sydney launched its first China Learning Centre (CLC) in T3 2020 at the Yixing Industrial Park for Environmental Science and Technology (ES&TP), where UNSW Centre for Transformational Environmental Technologies (CTET) is located, to support those students who cannot return to the campus in Sydney to continue their studies. From T3 2020 to T1 2022, the Yixing CLC has assisted more than 420 students to take online courses, develop thesis projects and carry out industrial training.
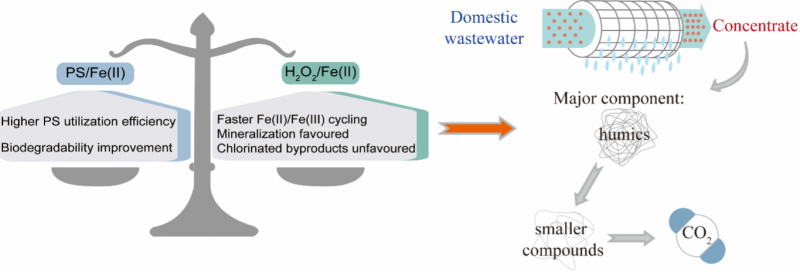 1. CTET recently published a research paper entitled “Comparative Experimental and Computational Studies of Hydroxyl and Sulfate-radical Mediated Degradation of Simple and Complex Organic Substrates” in Environmental Science and Technology. The paper builds on the outcome of a cooperating project between CTET and Beijing Originwater Technology Co., Ltd, which is aimed at treating nanofiltration concentrates from a municipal wastewater treatment plant. Through a systematic comparison between the hydroxyl and sulfate-radical mediated advanced oxidation processes, this work articulates the strengths and weaknesses of the two processes for application to wastewater treatment. Results of the study suggest that the sulfate-radical mediated process is not suitable for treatment of wastewaters containing high concentrations of organic contaminants and halide ions in view of the long -time scales required for oxidation of the organic contaminants, the excessive increase in concentration of SO42− in the treated wastewater and the possible formation of chlorinated by-products.
1. CTET recently published a research paper entitled “Comparative Experimental and Computational Studies of Hydroxyl and Sulfate-radical Mediated Degradation of Simple and Complex Organic Substrates” in Environmental Science and Technology. The paper builds on the outcome of a cooperating project between CTET and Beijing Originwater Technology Co., Ltd, which is aimed at treating nanofiltration concentrates from a municipal wastewater treatment plant. Through a systematic comparison between the hydroxyl and sulfate-radical mediated advanced oxidation processes, this work articulates the strengths and weaknesses of the two processes for application to wastewater treatment. Results of the study suggest that the sulfate-radical mediated process is not suitable for treatment of wastewaters containing high concentrations of organic contaminants and halide ions in view of the long -time scales required for oxidation of the organic contaminants, the excessive increase in concentration of SO42− in the treated wastewater and the possible formation of chlorinated by-products.